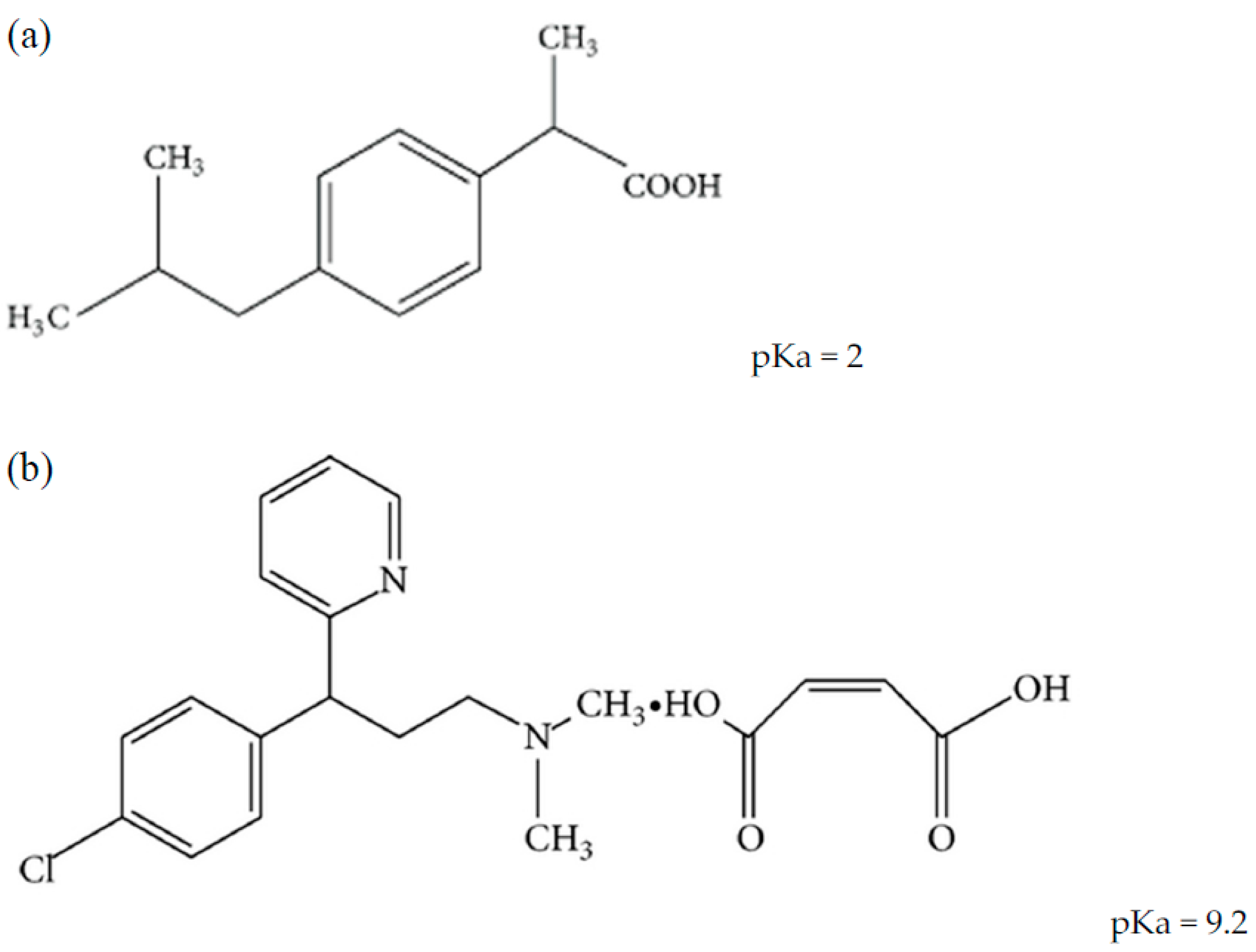
Universal Trends between Acid Dissociation Constants in Protic and Aprotic Solvents - Busch - 2022 - Chemistry – A European Journal - Wiley Online Library

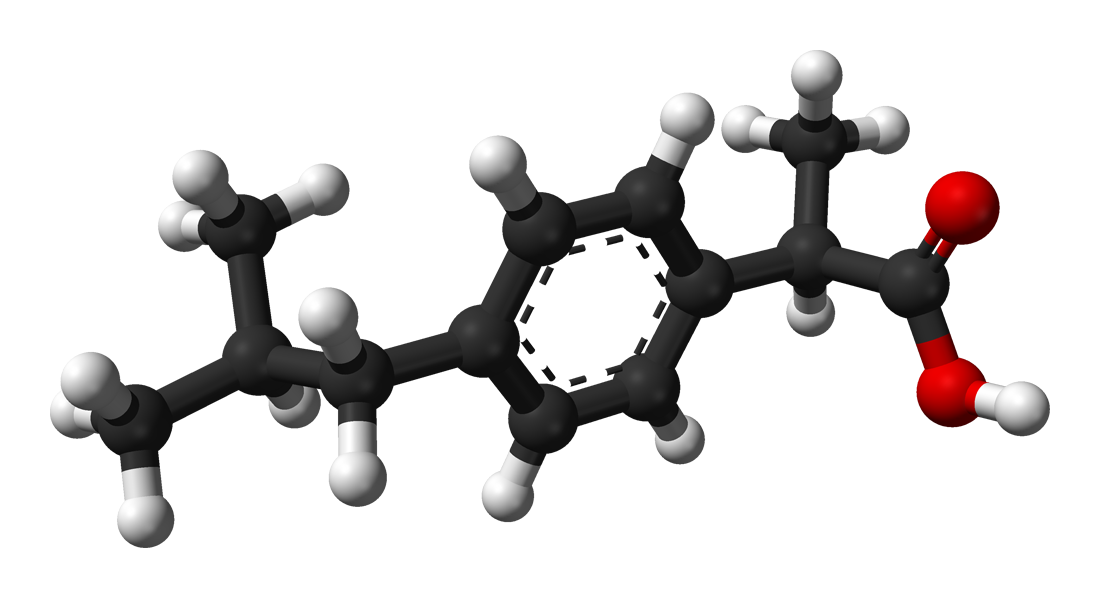
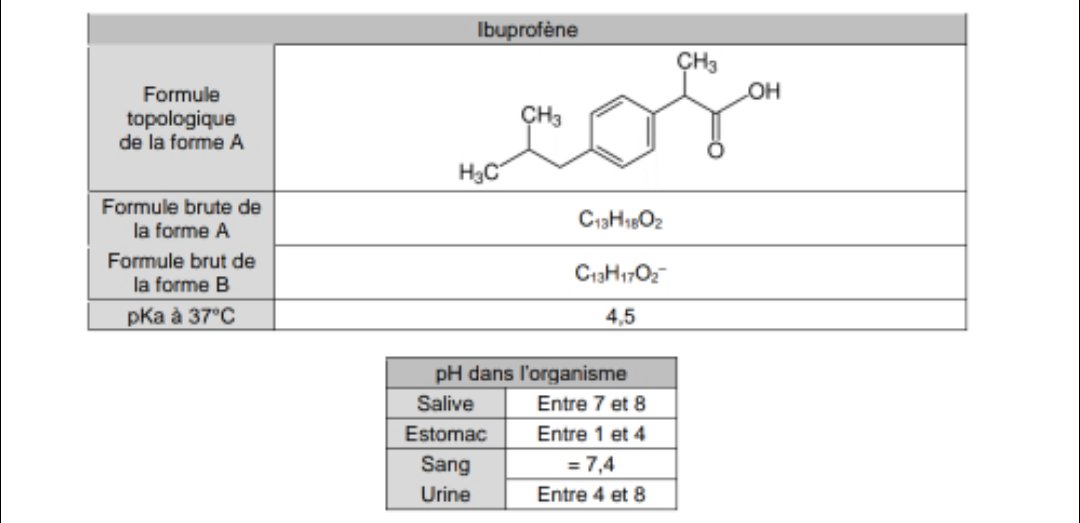
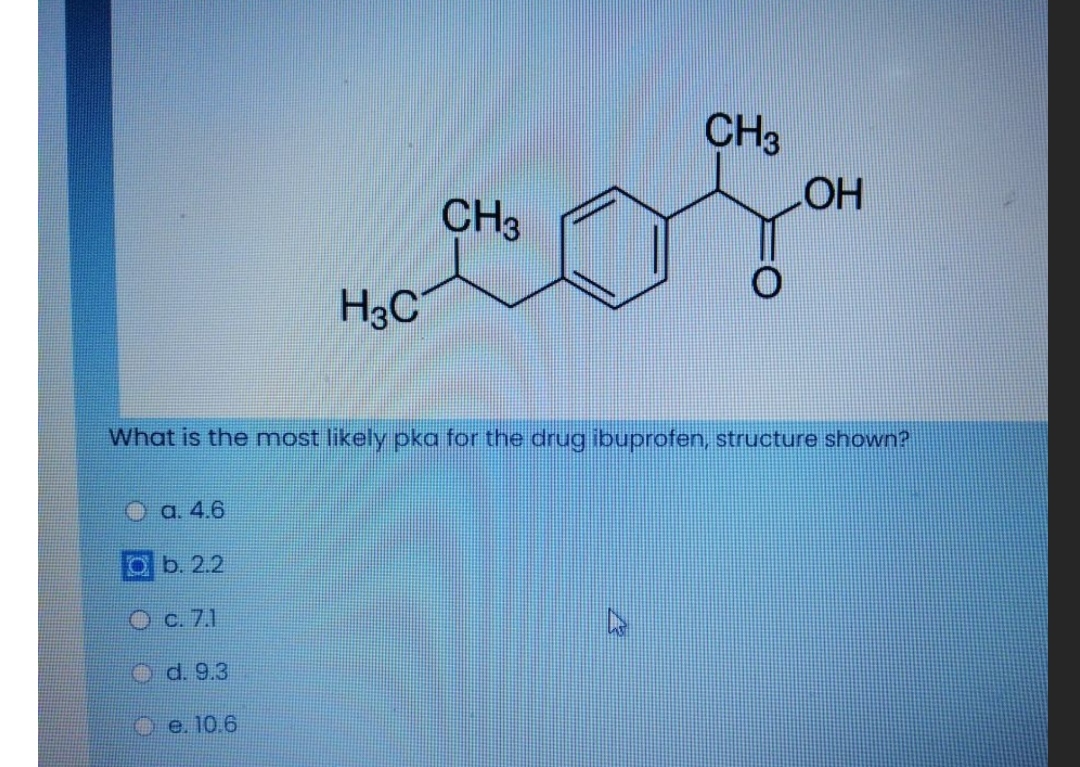
Figure1: Ibuprofen with a molecular weight of 206.3, pKa of 4.9, and... | Download Scientific Diagram
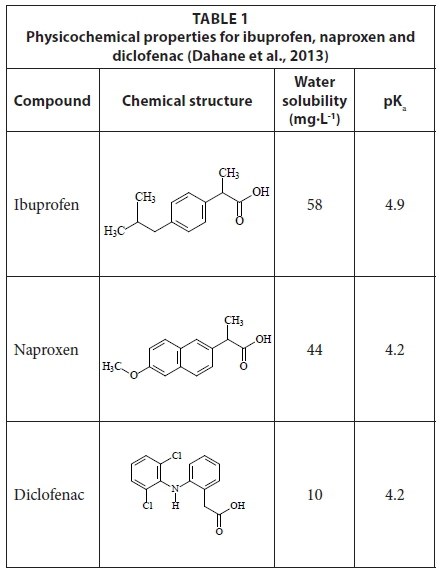
Simultaneous determination of naproxen, ibuprofen and diclofenac in wastewater using solid-phase extraction with high performance liquid chromatography

Multiple binding modes of ibuprofen in human serum albumin identified by absolute binding free energy calculations | bioRxiv
Sci. Pharm. | Free Full-Text | Development of HPLC Method for Simultaneous Determination of Ibuprofen and Chlorpheniramine Maleate
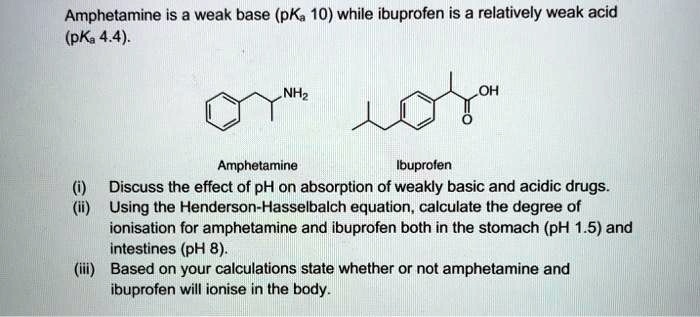
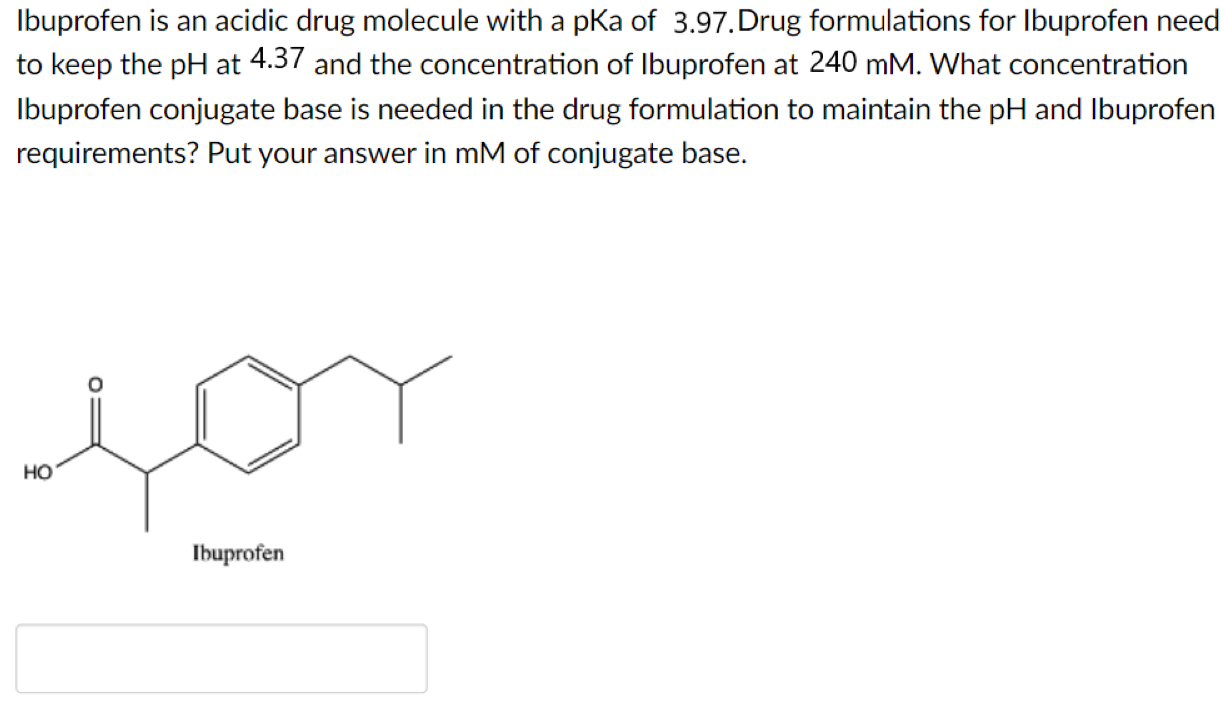
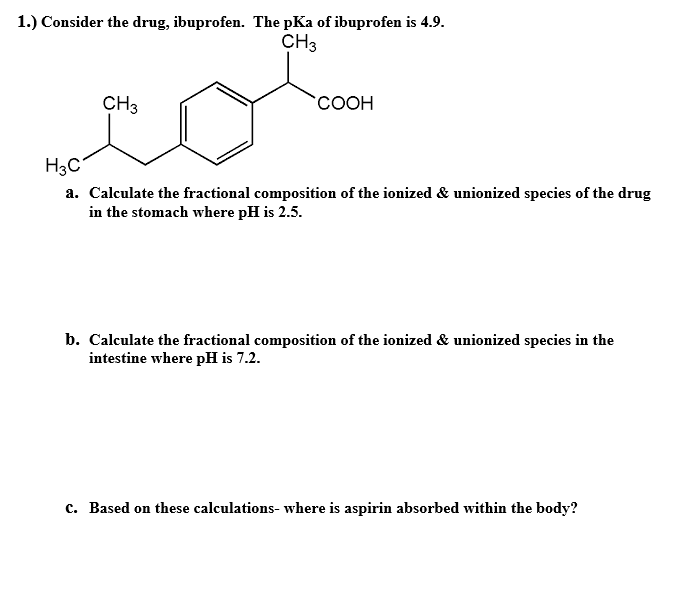
SOLVED: Amphetamine is a weak base (pKa 10) while ibuprofen is a relatively weak acid (pKa 4.4). Discuss the effect of pH on absorption of weakly basic and acidic drugs. Using the
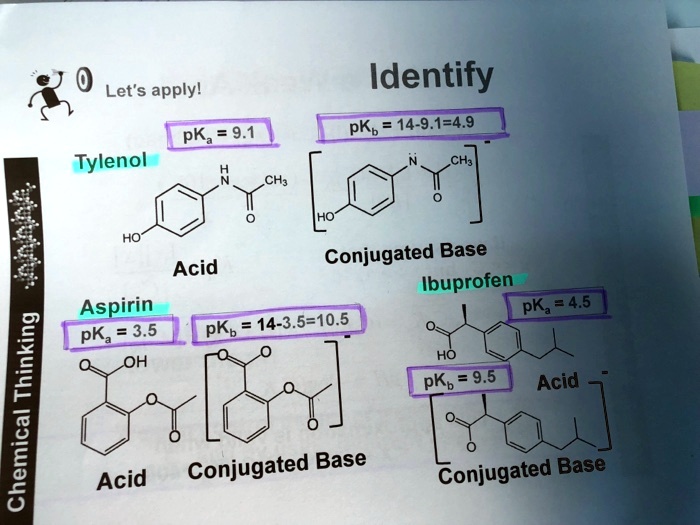
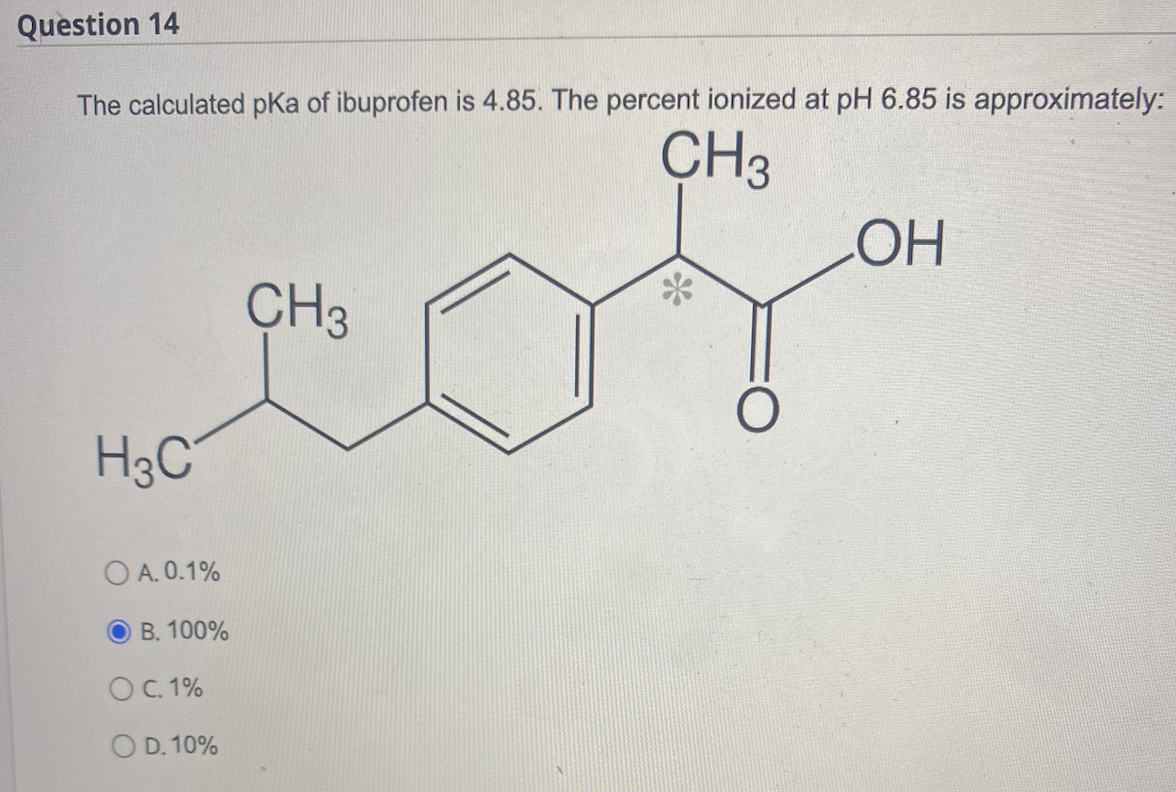
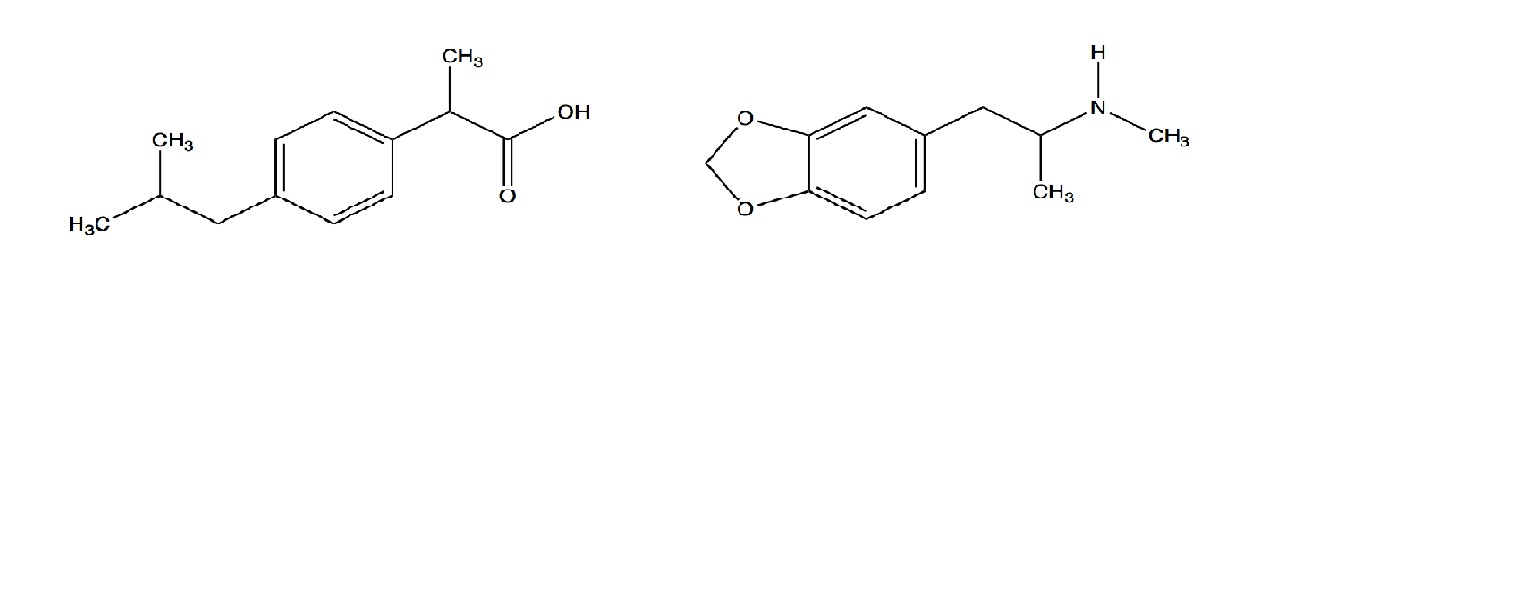
SOLVED: Let's apply. Identify pKb = 14 - 9.1 = 4.9. pKa = 9.1. Tylenol. CH3. CH3. HO. Conjugated Base Acid Ibuprofen Aspirin pKa = 4.5 pKb = 14 - 3.5 - 10.5 pKa OH HO pKb = 9.5 Acid. 1 7 Acid Conjugated Base Conjugated Base.

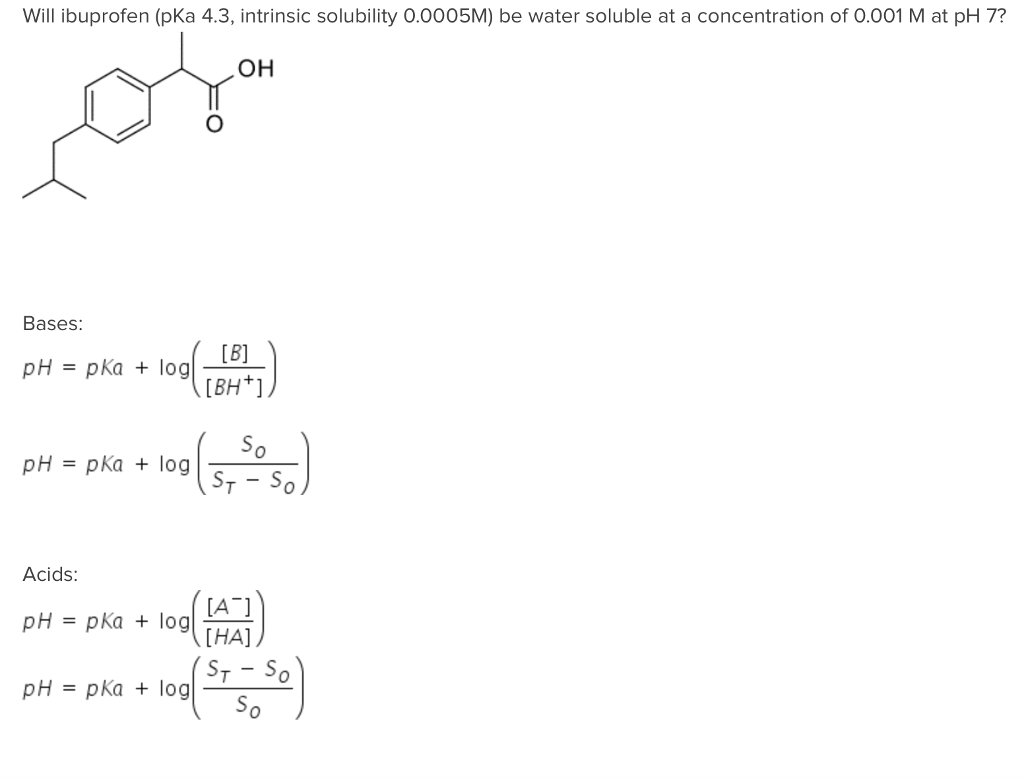
Ibuprofen: water affinity, effect of acidic pH and resonance structure:... | Download Scientific Diagram